Mostrar el registro sencillo del ítem
dc.contributor.author
de Tullio, Luisina

dc.contributor.author
Kaniecki, Kyle
dc.contributor.author
Kwon, Youngho
dc.contributor.author
Crickard, J. Brooks
dc.contributor.author
Sung, Patrick
dc.contributor.author
Greene, Eric C.
dc.date.available
2018-08-29T18:05:15Z
dc.date.issued
2017-10
dc.identifier.citation
de Tullio, Luisina; Kaniecki, Kyle; Kwon, Youngho; Crickard, J. Brooks; Sung, Patrick; et al.; Yeast Srs2 helicase promotes redistribution of single-stranded DNA-Bound RPA and Rad52 in homologous recombination regulation; Elsevier B.V.; Cell Reports; 21; 3; 10-2017; 570-577
dc.identifier.issn
2211-1247
dc.identifier.uri
http://hdl.handle.net/11336/57542
dc.description.abstract
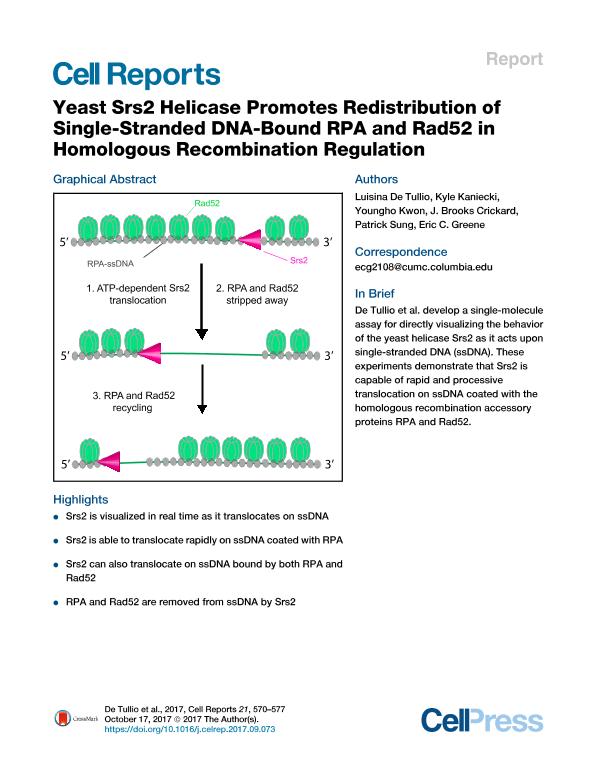
Srs2 is a super-family 1 helicase that promotes genome stability by dismantling toxic DNA recombination intermediates. However, the mechanisms by which Srs2 remodels or resolves recombination intermediates remain poorly understood. Here, single-molecule imaging is used to visualize Srs2 in real time as it acts on single-stranded DNA (ssDNA) bound by protein factors that function in recombination. We demonstrate that Srs2 is highly processive and translocates rapidly (∼170 nt per second) in the 3′→5′ direction along ssDNA saturated with replication protein A (RPA). We show that RPA is evicted from DNA during the passage of Srs2. Remarkably, Srs2 also readily removes the recombination mediator Rad52 from RPA-ssDNA and, in doing so, promotes rapid redistribution of both Rad52 and RPA. These findings have important mechanistic implications for understanding how Srs2 and related nucleic acid motor proteins resolve potentially pathogenic nucleoprotein intermediates. De Tullio et al. develop a single-molecule assay for directly visualizing the behavior of the yeast helicase Srs2 as it acts upon single-stranded DNA (ssDNA). These experiments demonstrate that Srs2 is capable of rapid and processive translocation on ssDNA coated with the homologous recombination accessory proteins RPA and Rad52.
dc.format
application/pdf
dc.language.iso
eng
dc.publisher
Elsevier B.V.
dc.rights
info:eu-repo/semantics/openAccess
dc.rights.uri
https://creativecommons.org/licenses/by-nc-sa/2.5/ar/
dc.subject
Dna Curtain
dc.subject
Homologous Recombination
dc.subject
Rad52
dc.subject
Replication Protein A
dc.subject
Single Molecule
dc.subject
Srs2
dc.subject.classification
Otras Ciencias Biológicas

dc.subject.classification
Ciencias Biológicas

dc.subject.classification
CIENCIAS NATURALES Y EXACTAS

dc.title
Yeast Srs2 helicase promotes redistribution of single-stranded DNA-Bound RPA and Rad52 in homologous recombination regulation
dc.type
info:eu-repo/semantics/article
dc.type
info:ar-repo/semantics/artículo
dc.type
info:eu-repo/semantics/publishedVersion
dc.date.updated
2018-08-27T18:58:33Z
dc.identifier.eissn
2211-1247
dc.journal.volume
21
dc.journal.number
3
dc.journal.pagination
570-577
dc.journal.pais
Estados Unidos

dc.description.fil
Fil: de Tullio, Luisina. Columbia University In The City Of New York; . Consejo Nacional de Investigaciones Científicas y Técnicas. Centro Científico Tecnológico Conicet - Córdoba. Centro de Investigaciones en Química Biológica de Córdoba. Universidad Nacional de Córdoba. Facultad de Ciencias Químicas. Centro de Investigaciones en Química Biológica de Córdoba; Argentina
dc.description.fil
Fil: Kaniecki, Kyle. Columbia University; Estados Unidos
dc.description.fil
Fil: Kwon, Youngho. University of Yale. School of Medicine; Estados Unidos
dc.description.fil
Fil: Crickard, J. Brooks. Columbia University; Estados Unidos
dc.description.fil
Fil: Sung, Patrick. University of Yale. School of Medicine; Estados Unidos
dc.description.fil
Fil: Greene, Eric C.. Columbia University; Estados Unidos
dc.journal.title
Cell Reports
dc.relation.alternativeid
info:eu-repo/semantics/altIdentifier/url/https://www.ncbi.nlm.nih.gov/pubmed/29045827
dc.relation.alternativeid
info:eu-repo/semantics/altIdentifier/doi/http://dx.doi.org/10.1016/j.celrep.2017.09.073
Archivos asociados